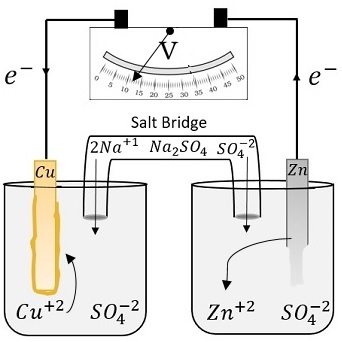
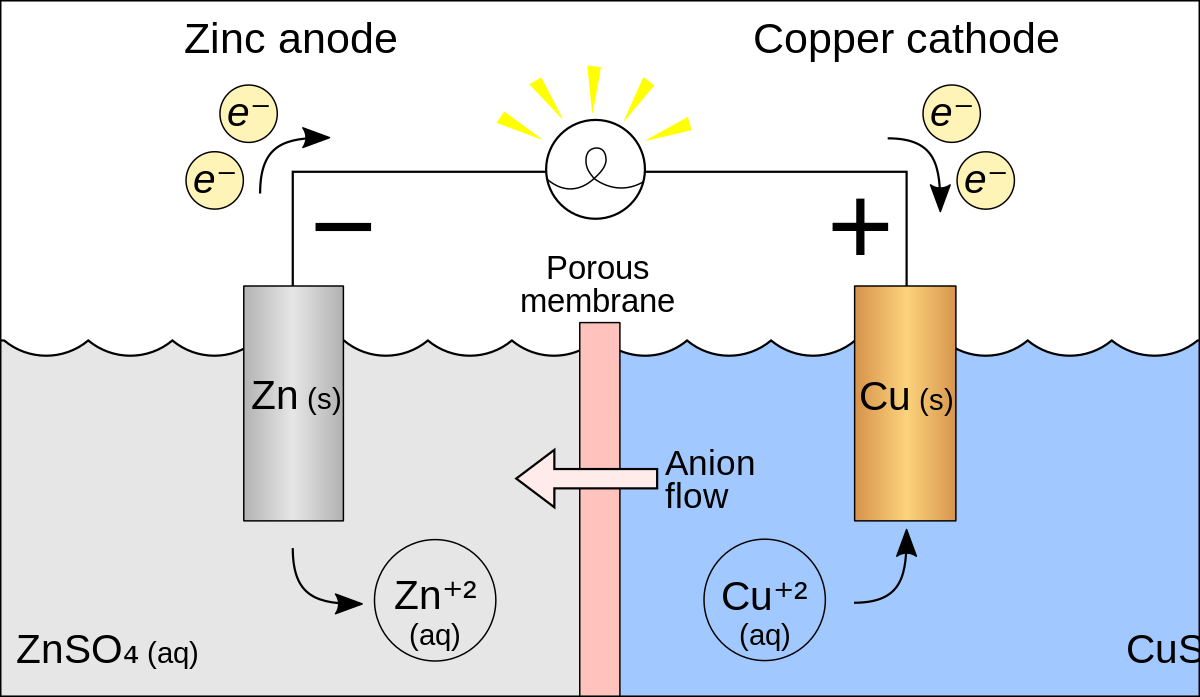
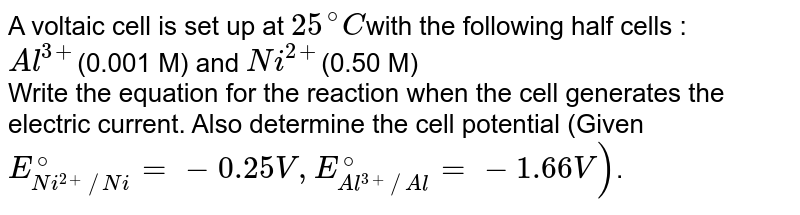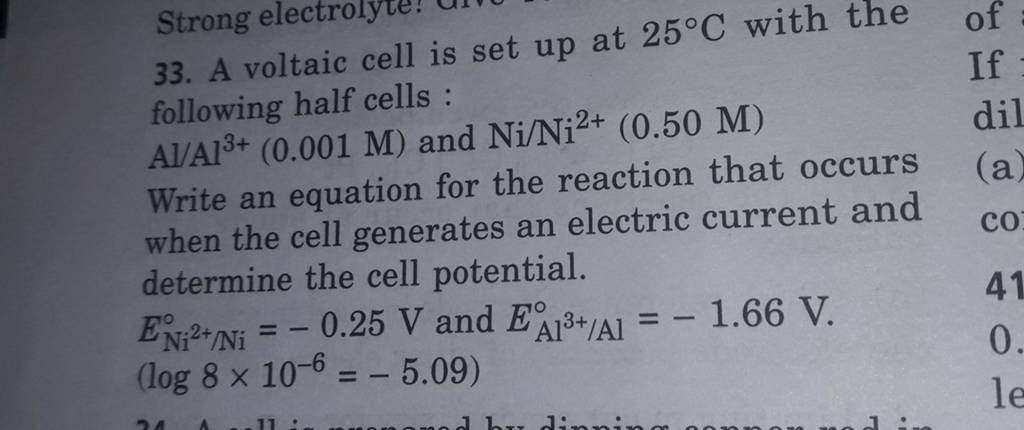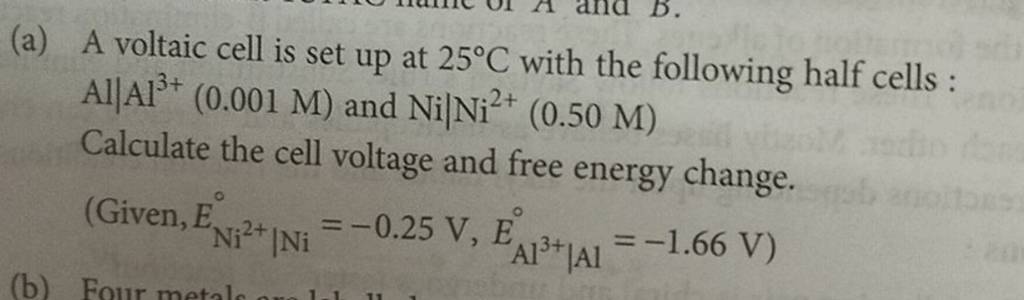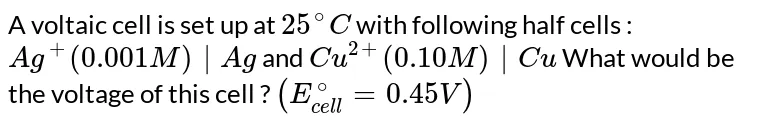A voltaic cell is setup at 25°C with the half cells Ag^+ (0.001 M) Ag and Cu^2+ (0.10 M) Cu. What should be its cell potential ? - Sarthaks eConnect | Largest Online Education Community

taic cell is up 25 °C with the following half cells, 9. 20. A voltaic cell is ur A bovlofti eroinsta A13+ and NiNi2+ (0.001 M) (0.5 M) EX13+ / Al = -
A voltaic cell is set up at 25°C with the following half cells. Al | Al^3+ (0.0010 M) and Ni^2+ (0.50 M) | Ni - Sarthaks eConnect | Largest Online Education Community
A voltaic cell is set up 25° C with the following half-cells: - Sarthaks eConnect | Largest Online Education Community

A voltaic cell is set-up 25°C with the following half-cells. Ag (0.001 M) | Ag and Cu²+(0.10 M) | Cu What would be the voltage of this cell? [Given, Ecell = 0.46
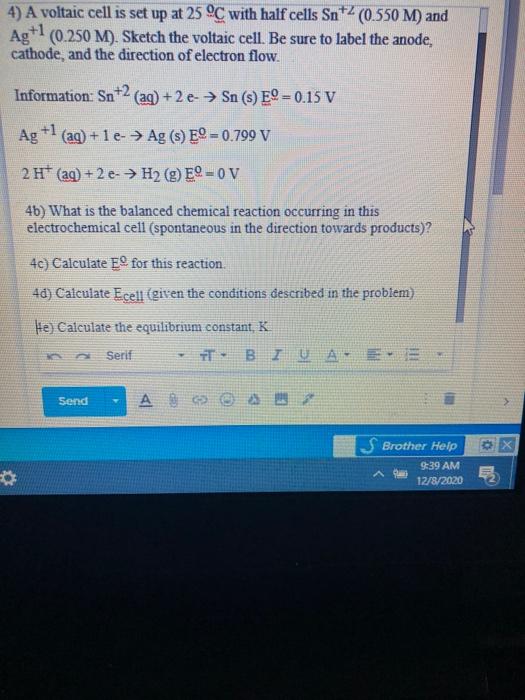
Please answer this Q1 A voltaic cell is set up at 25 C With following 12 - Chemistry - Electrochemistry - 13799617 | Meritnation.com

A voltaic cell in set up at 25^oC with the following half cells: Al/Al^3 + (0.001 M) and Ni/Ni^2 + (0.50 M) Write an equation for the reaction that occurs when the
A copper_ silver cell is set up. The copper ion concentration is 0.10 M. The concentration of silver ion is not know.the cell potential when measured was 0.422V determine the concentration of